Our Approach
The Fortier group specializes in the synthesis, purification, crystallization, handling, and characterization of air- and water-sensitive molecules and paramagnetic complexes. We use a combination of synthetic inorganic chemistry, X-ray crystallography, spectroscopy, and computational methods to understand the electronic structures and reactivity of novel metal complexes.

Low-Valent Metal Chemistry
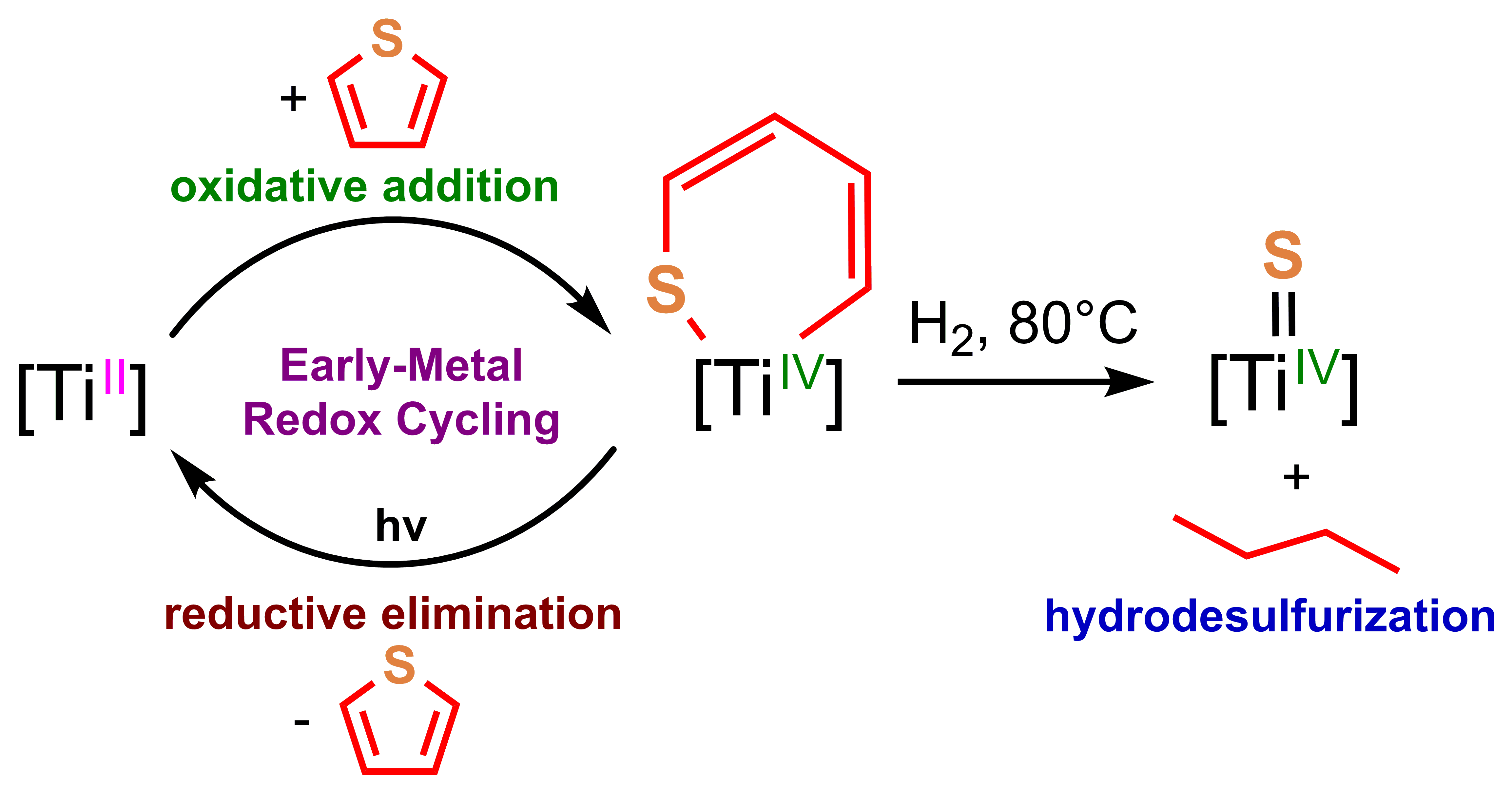
A central theme of our research is the synthesis and study of metals in unusually reduced oxidation states, which give rise to highly reactive species. A particular focus is the reduction of highly electropositive elements such as the early-metals and f-elements, where the thermodynamic driving force can generate potent reductants capable of multi-electron transfer. In this context, we have synthesized "molecular capacitors" capable of 8-electron discharge. These supercharged metal complexes show promise for base metal redox catalysis involving two-electron redox cycling, a feature typically associated with precious metals.
Our group has developed novel synthetic strategies for accessing and stabilizing low-valent complexes of titanium, uranium, and other metals, and we use a combination of X-ray crystallography, spectroscopy, and computation to characterize their structure and reactivity.
Representative topics:
- Synthesis of low-valent titanium synthons
- Redox chemistry of highly reduced metal complexes
- C–H and small molecule activation
- Uranium arene and sandwich complexes
Uranium and F-element Science
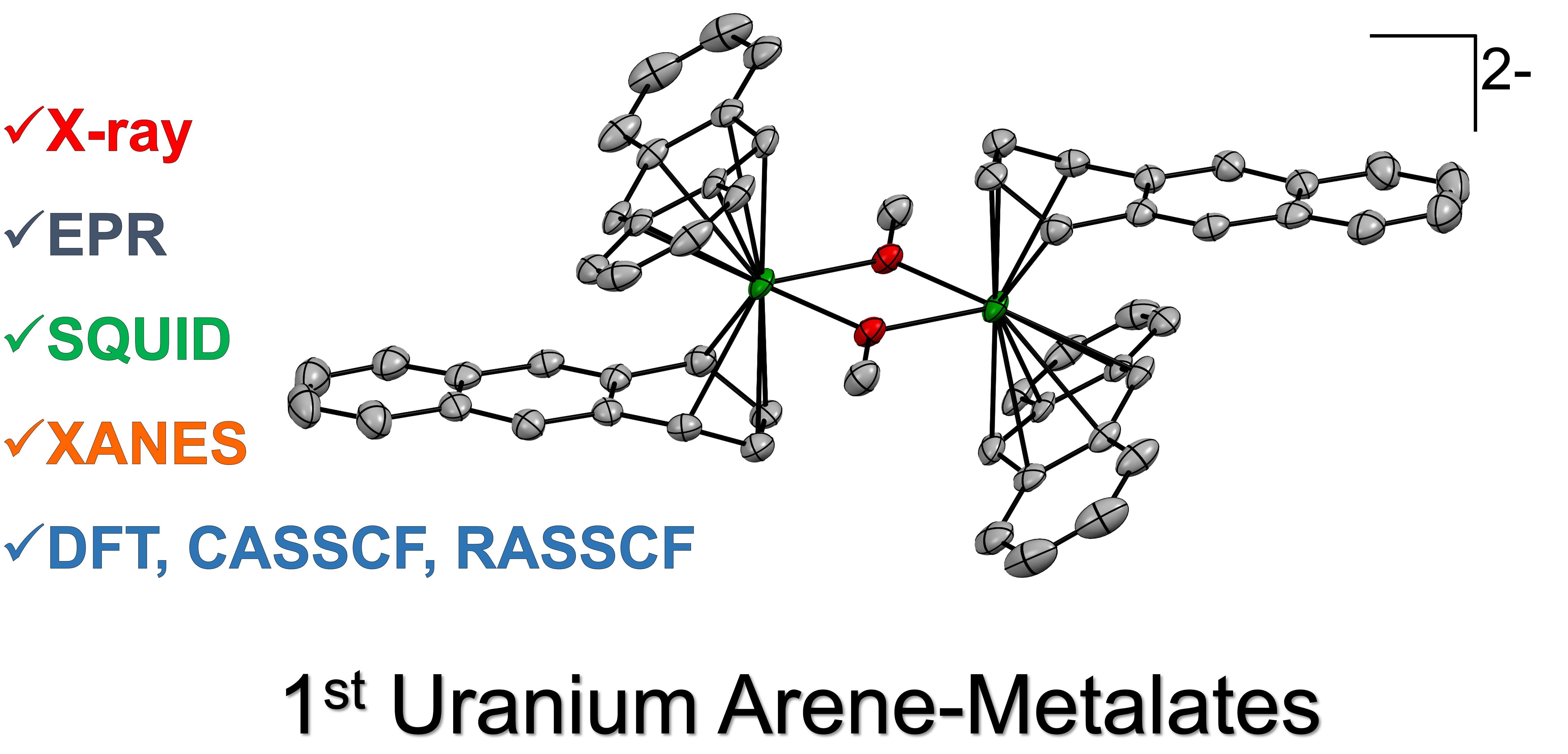
The f-block elements exhibit chemistry that is distinct from transition metals, owing to the unique spatial and energetic properties of f-orbitals. We investigate the structure, bonding, and reactivity of uranium and other f-element complexes to better understand the fundamental principles that govern actinide and lanthanide chemistry. In the actinides, their unique 5f and 6d/7s/7p valence orbital combinations hold potential for reactivity patterns that are, in principle, inaccessible to transition metals, making this an important frontier for both fundamental chemical understanding and reactivity discovery.
Our group has made contributions to uranium carbene, nitride, and arene chemistry, including the first examples of unsupported uranium arenide sandwich complexes.
Representative topics:
- Study of uranium-carbon bonding
- Comparative f-block chemistry (Ce, U, Np, Pu)
- XAS and advanced spectroscopy
- Highly reduced arene complexes
Ligand Design
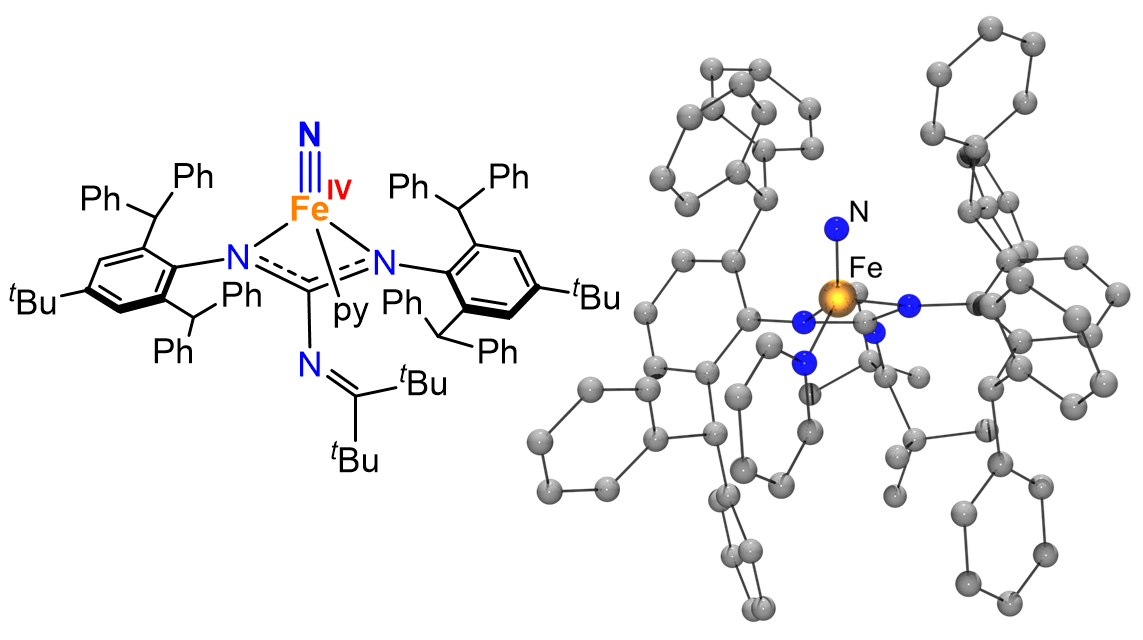
Enabling access to highly reactive and unusual metal complexes requires careful design of the ligand environment. We design and synthesize sterically demanding and electronically tunable ancillary ligands that can stabilize reactive metal centers and enforce unusual coordination geometries and oxidation states.
Our ligand design efforts are tightly integrated with our synthetic goals, allowing us to access metal complexes that may otherwise be too reactive to isolate and study.
Representative topics:
- Super bulky guanidinate and amide ligands
- Classic and non-classic redox-active ligand platforms
- Supporting ligands for low-coordinate metal complexes